|
3/8/2024 0 Comments Ansi asq z1 4 2003 r2018 pdf  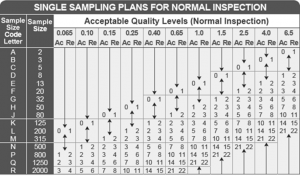 
Sampling Procedures and Tables for Inspection by Attributes for inspection of product’s or item’s flaws. This practice is widespread in pharmaceutics to adopt American National Standards Institute (ANSI)/American Society for Quality (ASQ) Z1.4-2008. There are attribute sampling plans that are primarily utilized to examine the productiveness of an item, product, or method to decide the compliance rate with already existing criteria. Please read this information until the end without skipping anything to understand better these terms and what they imply. At least the blur lines between these concepts will be cleared. By the end of this article, we hope you will have a firm knowledge of the topic. This article will let you know all the relevant things related to this topic and the significant differences between them. In case you are one of such people, then you have landed on the right platform. So they have to go anywhere else searching for the correct information. We have done some extensive research for our user’s ease. The concepts of ANSI ASQ Z1.4 AQL might probably seem complicated to some, but there must be others who have some idea of what they refer to and what it is all about. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
